 |
 Medical Device Engineering Services
» Medical Device Engineering
» International Projects » Project Management » Consulting Programs » Expert Witness & Analysis Engineering
Disciplines
Regulatory Documentation Services
|
|
 |
Copyright 2006 © Toltec International, Inc. All rights reserved.
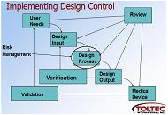 •
•